I had an unusual morning, talking with an AI about a topic I’ve long considered a personal failure.
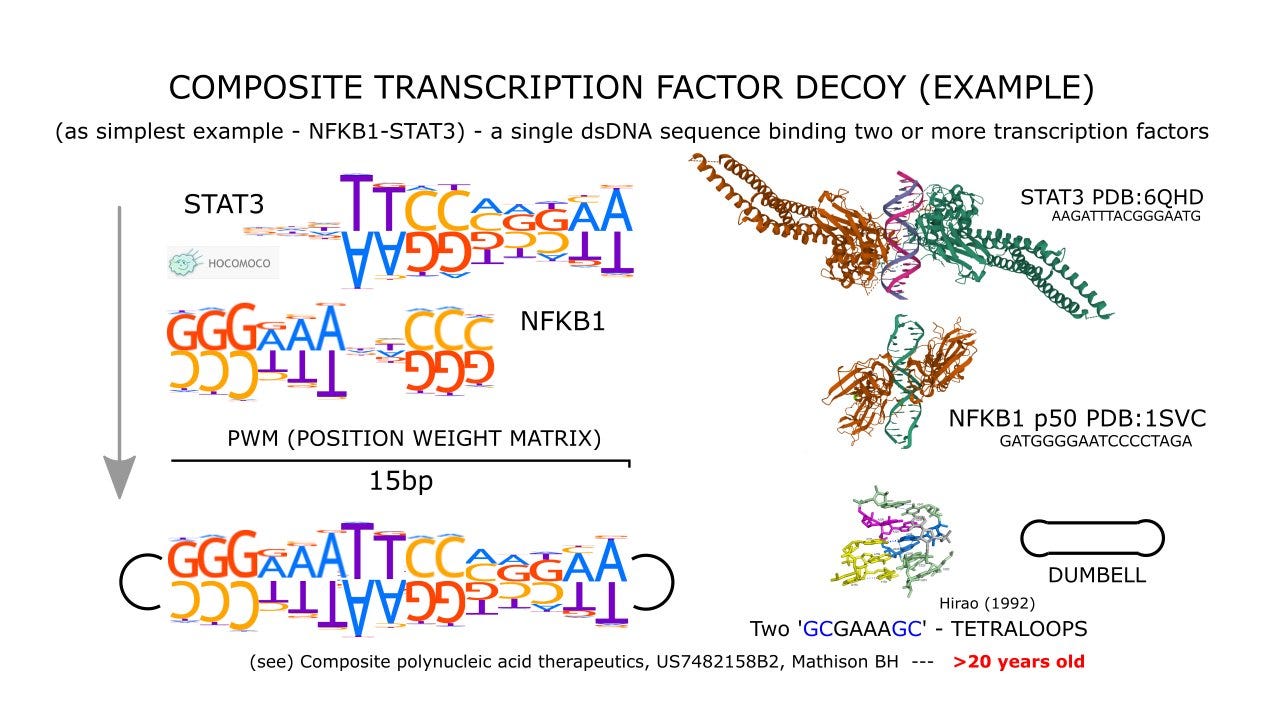
My background is in toxicology, with a specialization in genetic toxicology and chemical carcinogenesis, but my deepest scientific interests expanded into a different but related area of nucleic acid chemistry and therapeutics. More than twenty years ago, after working with an antisense oligonucleotide and RNA interference company, I began exploring the concept of designing cancer therapeutics in the related area of transcription factor decoys.1
This needs some backstory so consider a few scientific publications defining cancer hallmarks2 that seem to expand each decade.3 4 I am more of a splitter, but to be a lumper for a moment, consider two cancer aspects as being super simple: (1) unregulated ‘new’ cellular proliferation (e.g., neoplasia), and (2) the ability to spread (e.g., metastasize5). These two aspects are supported by networks involving genetic and epigenetic alterations of interconnected signaling pathways and transcriptional networks. Let me just say that trying to reduce cancers to a fixed set of static hallmarks might be a touch misguided.6 That said, targeting key transcription factors may be one approach to therapeutics,7 and doing so simultaneously with what I describe as a composite polynucleotides could offer a better strategy to disrupt the crosstalk that sustains these malignant programs.8
So, did you follow all of that? Probably not and I wouldn’t blame you as it wasn’t exactly clear. And that’s just a glimpse into one of the ways I’ve stumbled, and kept stumbling, over the years. “Dumb it down,” people have told me. Easier said than done. So, this morning, on my usual walk, I decided to put that advice to the test in my own way. I asked AI about progress in the area I once championed and studied deeply. The conversation naturally drifted into familiar territory and this again being why this approach has struggled to gain traction. As I walked down the trail, thinking through these complex ideas, I remembered something a few of my mentors had said: that science doesn’t speak for itself; it needs a story. Sometimes you must reshape the narrative, make it accessible, even a little “business friendly.”
That clicked instantly with my recent San Diego trip and conversations with a few folks trying to secure funding for ambitious complicated project. Inspired, I challenged the AI: explain the therapeutic concept I once promoted, but this time I asked - use a metaphor. And just like that, the abstract ideas changed in some interesting ways. The first story it offered was militaristic. The second story used music. Wow, how did this ‘entity’ know my history in music? I then asked it to merge the two metaphorical spins, and what it gave me was this short story.9
The Castle
Imagine cancer as a sprawling, heavily fortified castle, its towering walls built from legions of mutated cells—each one learning, adapting, and strengthening with every assault. Inside its ramparts, a cabal of rogue proteins played a relentless, discordant symphony, their chaotic music driving uncontrolled growth, feeding the fires of inflammation, and fueling the castle’s expansion into new territories.
The kingdom’s healers had long sought an “Achilles’ heel,” a single crack in the stone, but every time they thought they’d found it, the castle adapted, sealing the breach before the attack could succeed.
Then came the composite polynucleotides—an elite coalition of specialized agents, each with a different skill. Some infiltrated the castle and silenced the musicians, cutting off the signals that orchestrated its growth and spread. Others dismantled the armories, stripping the defenders of their ability to evolve. Still others sabotaged the communication lines between towers, disrupting the crosstalk that kept the entire fortress unified. Bit by bit, the once-impenetrable walls began to crumble. For the first time, the kingdom saw a clear path to victory.
Close
So, to close, this isn’t just wishful thinking. In cancer research, we know these cellular fortresses are expert adapters. Multiple transcription pathways and signaling networks are tangled together, keeping cells in a hyperactive, proliferative state—and that’s why hitting just one target rarely works.
Lately, the idea of hitting several pathways at once with drug combinations or engineered biologics is gaining traction. I’d like to think that decades-old patent I looked at this morning10 was ahead of its time in this regard, showing how multiple transcription factors tied to cancer and inflammation could be disrupted, breaking the crosstalk that makes tumors so stubborn.
So, what’s the takeaway? Sometimes the science is solid, the timing is perfect, and yet the story just doesn’t land. Still, I loved how this “castle” metaphor tied everything together, offering a glimpse of a possible step forward. It was pretty wild how neatly it captured the essence of a long, detailed conversation. Science—and storytelling—are definitely changing.
Let me add a few references in support of this idea.11
Stay safe out there…
A transcription factor decoy is a synthetic nucleic acid that imitates the specific DNA sequence a transcription factor would normally bind to in a gene’s promoter or enhancer region. These short, double-stranded oligonucleotides act as molecular mimics, binding to the transcription factor and preventing it from attaching to its natural genomic target. By competing for the transcription factor’s binding, the decoy can block or alter the expression of genes under its control. This approach has been used experimentally to modulate pathways involved in cancer, inflammation, and cardiovascular disease, with early studies showing that a decoy targeting the E2F transcription factor could prevent smooth muscle proliferation in injured arteries. Because transcription factors are difficult to target with conventional small molecules, decoys provide a way to directly interfere with the DNA-binding activity that drives their function, offering a versatile platform for therapeutic intervention.
Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000 Jan 7;100(1):57-70. doi: 10.1016/s0092-8674(00)81683-9. PMID: 10647931.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011 Mar 4;144(5):646-74. doi: 10.1016/j.cell.2011.02.013. PMID: 21376230.
Hanahan D. Hallmarks of Cancer: New Dimensions. Cancer Discov. 2022 Jan;12(1):31-46. doi: 10.1158/2159-8290.CD-21-1059. PMID: 35022204.
To spread from the primary site of origin to distant parts of the body, typically via the bloodstream or lymphatic system, forming secondary malignant growths (metastases) - from Greek metastasis, meaning “a change of place, removal, migration” (meta- = “beyond, after”; stasis = “placement, position”). A pathologist definition, to undergo metastasis; the process by which neoplastic (cancer) cells detach from the primary tumor, invade surrounding tissues, intravasate into the circulatory or lymphatic system, survive transit, extravasate at a distant site, and proliferate to form secondary malignant tumors (metastases). A very complex scientific topic. (I’m sill digesting this reference)
El-Tanani M, Rabbani SA, Babiker R, Rangraze I, Kapre S, Palakurthi SS, Alnuqaydan AM, Aljabali AA, Rizzo M, El-Tanani Y, Tambuwala MM. Unraveling the tumor microenvironment: Insights into cancer metastasis and therapeutic strategies. Cancer Lett. 2024 Jun 1;591:216894. doi: 10.1016/j.canlet.2024.216894. Epub 2024 Apr 16. PMID: 38626856.
Here is how A.I. might also expand this topic “Across roughly 2,600 whole-cancer genomes, most tumors contain a complex mix of point mutations, structural variants, and noncoding alterations that rewire regulatory circuitry, often through enhancer hijacking and changes in three-dimensional genome organization. These events operate alongside hundreds of potential oncogenes and tumor-suppressor genes, with their impact shaped by the cell of origin and the tissue context. Single-cell atlases reveal an even deeper layer of complexity, showing diverse malignant cell states and intricate tumor microenvironments, where lineage plasticity and epigenetic reprogramming drive disease progression and resistance to therapy. Noncoding RNAs—including miRNAs, lncRNAs, circRNAs, and piRNA-like pathways—as well as RNA-level processes such as splicing, editing, and alternative polyadenylation, further fine-tune these malignant programs. Distinct mutational processes, prominently APOBEC cytidine deamination, leave recognizable signatures across cancer genomes. Taken together, these findings underscore that cancer is best understood as a dynamic, multi-scale systems disease—one that spans the genome, epigenome, transcriptome, cell state, and ecosystem—rather than a fixed set of static hallmarks.” - ChatGPT-5
From a molecular toxicology perspective, defining the true number of distinct cancer types requires more than cataloging tissues of origin. One might begin by considering the diversity of stem cell populations and other cells of origin, then overlaying the spectrum of oncogenes and tumor-suppressor genes that can be altered within each. To this, we add the layers of coding and noncoding RNAs, along with the regulatory sequences and motifs that govern their expression. These in turn intersect with transcription factors and other DNA-binding proteins that shape and maintain differentiated cellular states—forming a complex, multi-tiered network in which malignant transformation can occur through numerous, context-dependent routes.
Khan AQ, Hasan A, Mir SS, Rashid K, Uddin S, Steinhoff M. Exploiting transcription factors to target EMT and cancer stem cells for tumor modulation and therapy. Semin Cancer Biol. 2024 May; 100:1-16. doi: 10.1016/j.semcancer.2024.03.002. Epub 2024 Mar 19. PMID: 38503384.
It bears repeating that cancers are highly adaptable biological systems. Through genetic mutations and epigenetic changes, they can reroute signaling pathways, bypass inhibitory checkpoints, and compensate for the loss of one proliferative mechanism by activating others. Many therapies, too many, act like precision arrows aimed at a single molecular target, only for the malignancies to evade through pathway redundancy or adaptive resistance. An effective strategy, I believed, would require a coordinated, multi-pronged assault aimed at disrupting several critical pathways simultaneously to overwhelm the tumor’s capacity to adapt.
What triggered this post :-)
US20060166916A1 - Composite polynucleic acid therapeutics - Google Patents
Let me quote myself from >20 years ago: “Highly proliferating cells and groups thereof are often the product of multiple events. With time and unchecked by normal processes maintaining homeostasis, proliferating cells expand in number where some may become genetically unstable and diverse. With continued growth, probabilities increase that cells within these groups acquire and express traits enabling escape from or adaptation to therapy. These cells and groups thereof with time will not possess a single target or Achilles' heel through which a mono-functional entity or “magic bullet” could achieve complete therapeutic success. When complex intracellular networks, crosstalk and cellular redundancies that maintain hyperproliferative states are considered, the discovery and development of mono-functional agents may not represent the best or only therapeutic option.” US20060166916A1 - Composite polynucleic acid therapeutics - Google Patents
Ji Z, He L, Regev A, Struhl K. Inflammatory regulatory network mediated by the joint action of NF-kB, STAT3, and AP-1 factors is involved in many human cancers. Proc Natl Acad Sci U S A. 2019 May 7;116(19):9453-9462. doi: 10.1073/pnas.1821068116. Epub 2019 Mar 25. PMID: 30910960; PMCID: PMC6511065.
Grivennikov SI, Karin M. Dangerous liaisons: STAT3 and NF-kappaB collaboration and crosstalk in cancer. Cytokine Growth Factor Rev. 2010 Feb;21(1):11-9. doi: 10.1016/j.cytogfr.2009.11.005. Epub 2009 Dec 16. PMID: 20018552; PMCID: PMC2834864.
Bushweller JH. Targeting transcription factors in cancer - from undruggable to reality. Nat Rev Cancer. 2019 Nov;19(11):611-624. doi: 10.1038/s41568-019-0196-7. Epub 2019 Sep 11. PMID: 31511663; PMCID: PMC8820243.









Share this post